
The transition or inner transition family forms the ionic chemical bond with metals (s-block) and covalent bond with metals non-metals (p-block). The name d-block (transition series) or f-block (inner transition series) on the periodic table is used due to the presence of progressively filled d or f-orbitals in the valence shell electronic structure. Since the second period of p block elements like boron, carbon, nitrogen, oxygen, fluorine, and argon have filled s-orbitals with valence shell electron configuration, 2s 2 2p 1→6, where n = number of period. Group-3, 4, 5, 6, 7, and noble gases belong to p-block elements. P-block on the periodic table organizes by progressively filled p-orbital in valence shell electronic structure but helium is an exception with electron arrangement 1s 2. The valence shell electron configuration, ns 1→2, where n = principle quantum number, or the number of periods. Group-1 (hydrogen, lithium, sodium, potassium, rubidium, cesium, and francium) and group-2 (beryllium, magnesium, calcium, strontium, barium, and radium) belong to s-block elements. 
The name s-block element in the periodic table is given due to the arrangement of electrons, the valence electron enters into ns-orbital and is filled progressively according to the configuration rules. The organization of the middle portion of the periodic table made the relationship between the left and right sections and contains a list of transition metals or d-block and inner transition or f-block elements.Īccording to the modern periodic law based on the atomic number and valence shell electron arrangement of elements, the different types of metals and non-metals in chemistry are organized to form s, p, d, and f-block on the periodic table.The right portion contains groups IIIA, IVA, VA, VIA, VIIA with high electron affinity, and non-reactive noble gases like helium, neon, argon, krypton, xenon, and radon.The left section in the table has group IA and IIA define the extremely high electropositive character but very low ionization energy and always show the positive oxidation number.Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous.According to the above definition and explanation, the long form of the periodic table is labeled into three units like left, right, and middle sections for learning chemistry or physics in science. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. 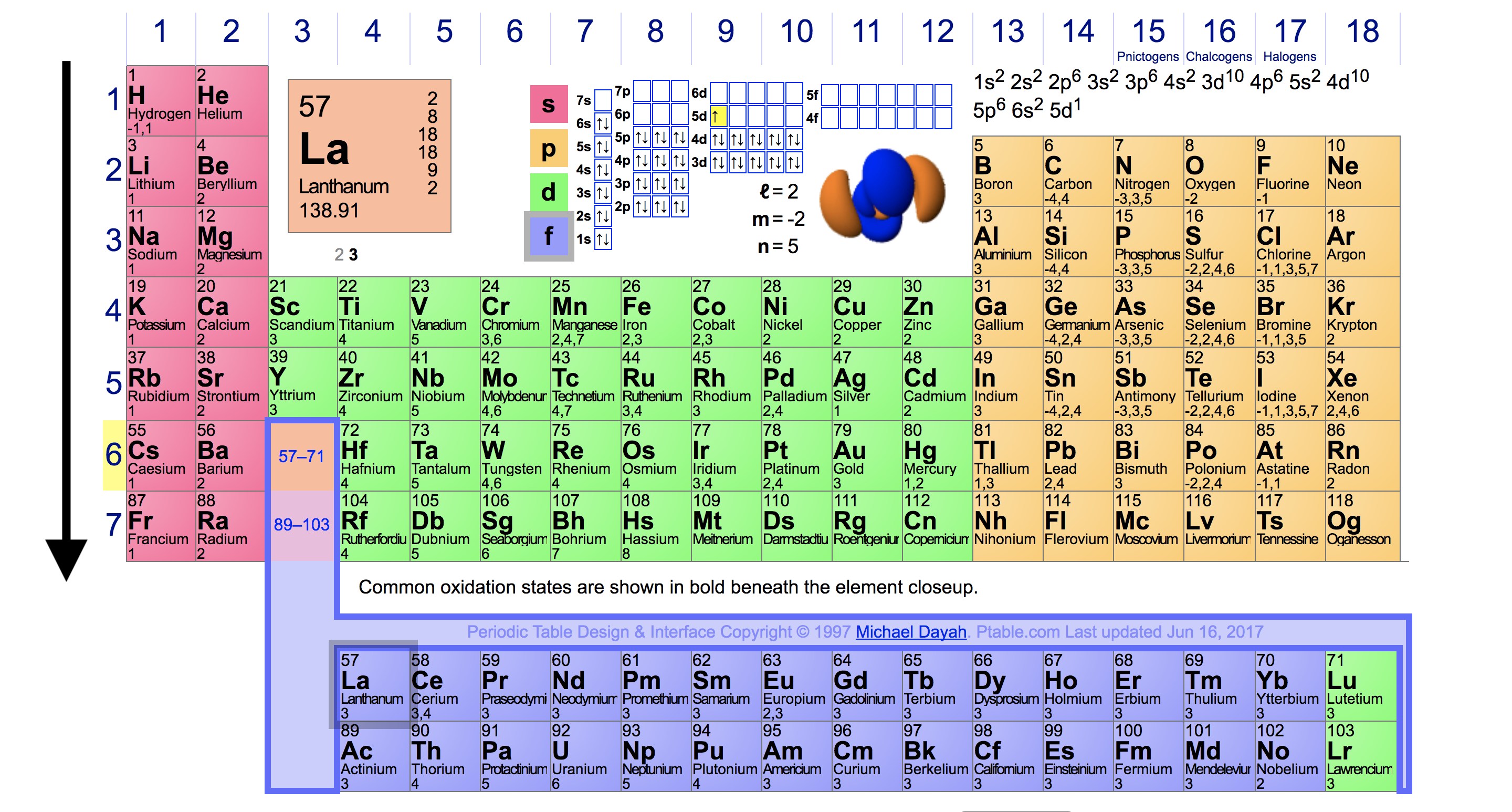
The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. 
\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
